Energy From Fuels
Chemical Energy \(=\) Potential Energy (Stored Energy) \(+\) Kinetic Energy (Energy of Movement)
Enthalpy refers to the chemical energy or heat content of a substance. The exchange of heat between a system and its surroundings under constant pressure is referred to as the enthalpy change
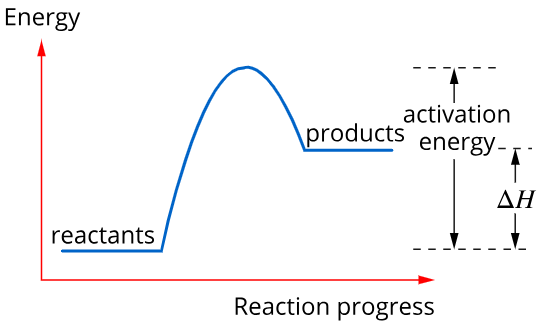
Exothermic reactions, e.g. combustion release energy into the surroundings. Therefore, the energy of the reactants is higher than the products and \(\Delta H=H<0\)
Exothermic Reaction
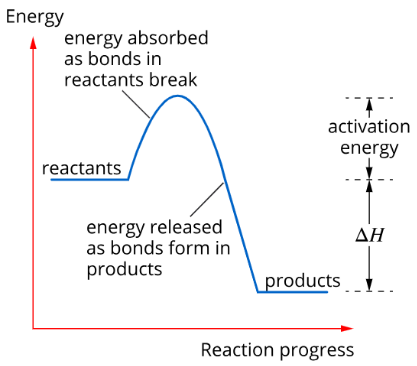
Endothermic reactions, e.g. cold packs, absorb energy from the surroundings. Therefore, the energy of the products is higher than the reactants and \(\Delta H=H>0\)
Endothermic Reaction
Activation energy is the energy required to break the bonds of the reactants so that the reaction can proceed.
These chemical reactions are merely those which include the \(\Delta H\) value and must include states.
For example: \(CH_4(g)+O_2(g)\rightarrow CO_2(g)+H_2O(l)\,\,\Delta H=882kJ\,mol^{-1}\)
Specific heat capacity is the amount of energy needed to increase the temperature of a substance by \(1^oC\)
\(q=m\times c\times \Delta T\)
The heat released or absorbed by a chemical substance during a change of state
\(q=n\times L\)
The heat combustion of a fuel is defined as the enthalpy change that occurs when a fuel burns completely in oxygen. However, as many fuels are mixtures of chemicals that don’t have a specific molar mass, they cannot be expressed in kJ/mol.
\(q=n\times \Delta H_C\)