Galvanic Cells
If the reactants come into direct contact, then their chemical energy is transformed directly to thermal energy. In a galvanic cell however, the half-reactions occur in separate containers, and therefore the electrons are transferred by an external circuit so that the chemical energy is transformed into electrical energy.
Galvanic cells are the simplest form of electrochemical cells, which convert chemical energy to electrical energy. The external circuit is the conductor for the electrons to flow through. The internal ciruit is the salt bridge, which contains an inert electrolyte to balance the charge in each half cell. Without it, the cell would become polarised as electrons accumulate in one half of the cell. Each galvanic cell consists of two half-cells, which contain an electrode i contact with a solution.
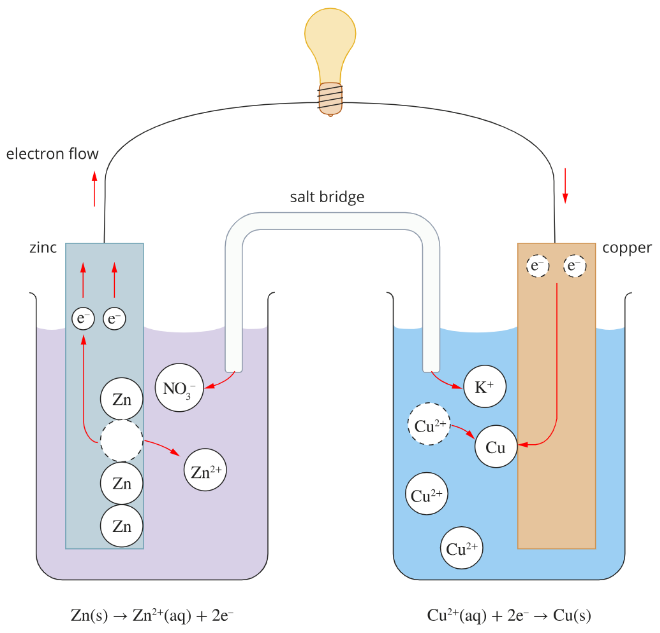
Galvanic Cell
The current in a galvanic cell flows because one half-cell has a greater tendency to push electrons into the external circuit than the other half-cell. The potential difference of a cell is denoted by \(E^o\) and is measured in volts. In a galvanic cell, the stronger reducing agent is with the anode, while the stronger oxidising agent is with the cathode. The \(E^o\) value can be found by subtracting the lower \(E^o\) from the higher \(E^o\) in the electrochemical series.
Note: This does not give any information about the rate at which reactions occur
For a spontaneous reaction to occur, a reaction that occurs naturally, the oxidising agent (on the left of the electrochemical series) must react with a reducing agent (on the right) that is lower in the series
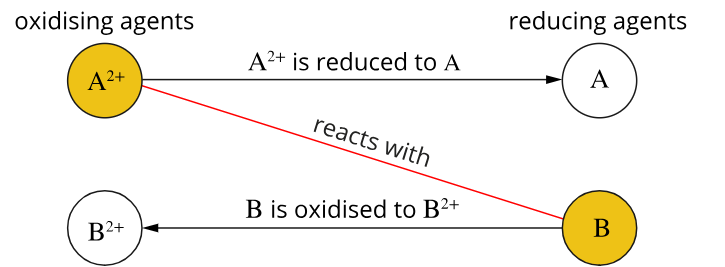
Spontaneous Reactions